Background
Alkene hydrosilylation is one of the prevalent methods for carbon-silicon bond formation in academic as well as in industrial settings. Usually catalyzed by precious late transition metal complexes, e.g. platinum, substantial progress is currently being made in the design of catalysts based on more abundant and therefore potentially less expensive transition metals. Various types of silanes are currently being employed in these catalyses.
However many of these silane catalysts like Me3SiH and Me2SiH2 are highly flammable and potentially explosive gases. Therefore the use of such catalysts is inconvenient from a safety point of view.
Up to now there are no practical methods capable of dealing with these safety issues. As silanes have a great potential within the hydrosilylation of alkenes, new methods for the use and/or the production of Hydrosilanes are needed.
Technical Description
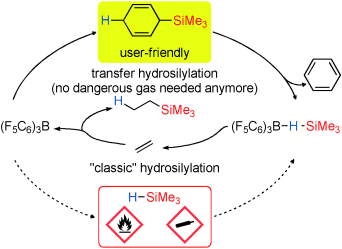
The invention from scientists of the TU Berlin relates to the use of cyclohexa-2,5-dien-1-yl-silanes as viable hydrosilane precursors. It is revealed that Lewis acids were able to catalyze the release of hydrosilanes from cyclohexa-2,5-dien-1-yl-silanes without the necessity to deal with the potential explosive gaseous phase.
Using the easy-to-handle 3-trimethylsilyl- or 3-dimethylsilylhexa-1,4-dienes 1 and 2, this method was succesfully applied to the in situ Generation of gaseous Me3SiH and Me2SiH2, silanes that are often prohibited from laboratories or safety considerations.
Possible Applications
The method according to the invention could make the use of hydrosilanes less dangerous and therefore more feasible.
Figure: Reaction scheme (A.Simonneau, M.Oestreich, Angew. Chemie, Volume 52, Issue 45, November 4, 2013, Pages 11905-11907).